Due to the nature of the product, the order and delivery of Osteocel Plus needs to be carefully coordinated. Osteocel Plus is not available for purchase online. Osteocel Plus requires Priority Shipping and Handling which is in addition to prices shown. Please call ACE SOUTHERN for ordering instructions.
Osteoconductivity: Mineralized cancellous scaffold
Osteoinductivity: MSC growth factor production, BMP production and demineralized cortical bone
Osteogenicity: Viable MSCs and osteoprogenitor cells
- Physiological: mimics biologic profile of autograft
- Consistent: each lot tested for cell concentration, cell viability, and cell activity (osteogenic potential)
Mesenchymal stem cells are immune privileged cells that do not stimulate a
cellular immune response. Osteocel Plus does not activate T cell proliferation,
as shown in vitro from Mixed Lymphocyte Reaction (MLR) testing. (1)
Histology from a human sinus augmentation study using Osteocel Plus shows
high vital bone content at 16 weeks, with very low residual graft material. (2)
MCSs contained in Osteocel® Plus are capable of differentiating into
bone-forming cells (osteoblasts). (3) Every lot of Osteocel Plus is tested
for bone-forming potential.
The osteogenic potential arises from the mesenchymal and osteoprogenitor
cells in Osteocel Plus. Following processing of marrow-rich bone, release
testing demonstrates osteogenic potential according to the following criteria:
- Rich supply of osteopotent cells: Greater than 250,000 cells/cc
- Viability: Greater than 70% cell viability
- Positive osteogenicity: In vitro cell culture assay
- Aseptic tissue processing
- Antimicrobial treatment of tissue
- Sterility cultures performed on every lot/bul
- Selective immunodepletion process
- No evidence of ectopic tissue formation or inflammatory cellular response with experimental high doses of MSCs cells
- Hepatitis B Surface Antigen/bul
- Hepatitis B Core Antibody • Hepatitis C Virus Antibody • Hepatitis C Nucleic Acid Test • HIV-1 and 2 Antibody • HIV-1 Nucleic Acid Test • Human T-Lymphotropic Virus Antibody I/II • Syphilis (Treponema Pallidum)
- Medical and social history evaluation
- Physical examination
- Medical record evaluation, including autopsy report (if performed)
- Licensed physician review and release of every donor record
- GTP compliant according to FDA regulations
- All applicable state licensure/ registrations
- AATB® Accreditation
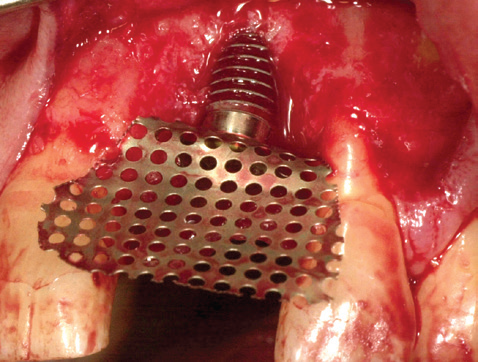
BEFORE: exposed implant
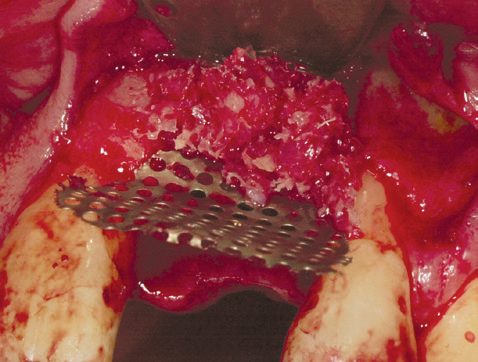
Bone graft in place prior to mesh fixation
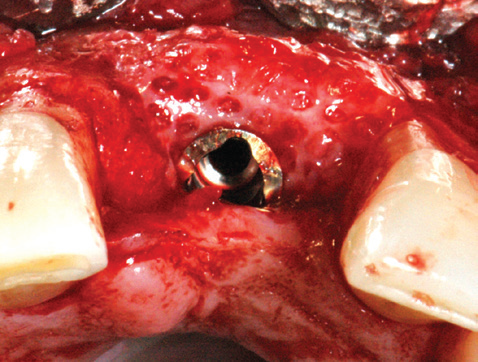
AFTER: 4 months after mesh removal

Upon obtaining Osteocel Plus from frozen storage, remove peel pack from protective covering. Note: Osteocel Plus cannot be refrozen.
Designated person opens sterile peel pack utilizing standard sterile technique.
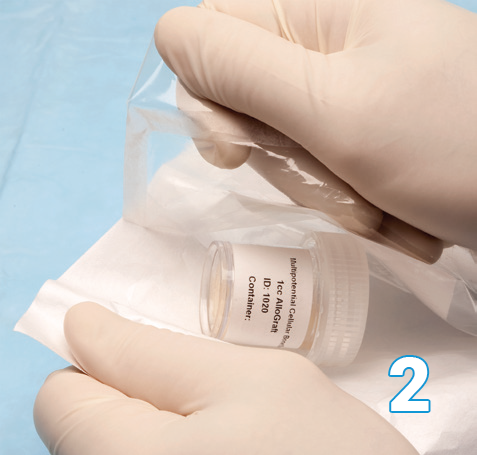
A sterile scrub person removes the Osteocel Plus container from the peel pack without compromising sterility.
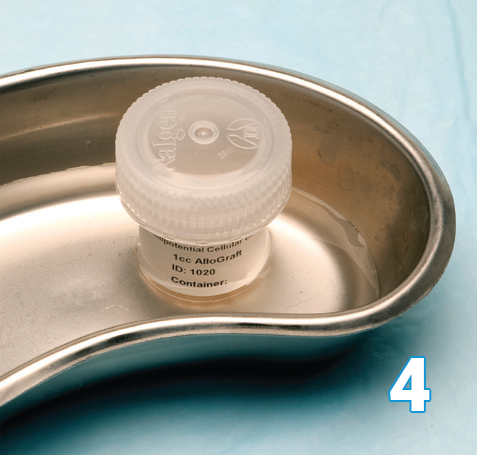
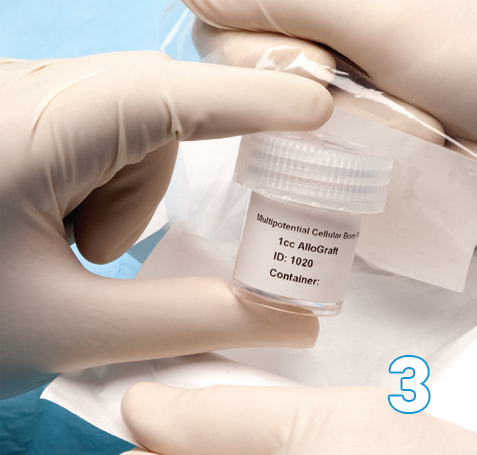
The Osteocel Plus container is placed into a basin of saline at physiological temperature (95-102°F / 35-39°C). Depending on size of container, Osteocel Plus will take 15-20 minutes to thaw. It can also be thawed at ambient temperature for 1 hour.
Note: Do not warm above 102°F/39°C.
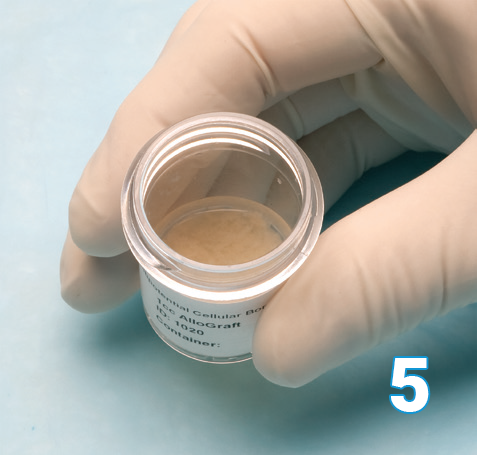
Once thawed Osteocel Plus will be at the bottom of the container with the cryopreservation cell saving liquid on the top.
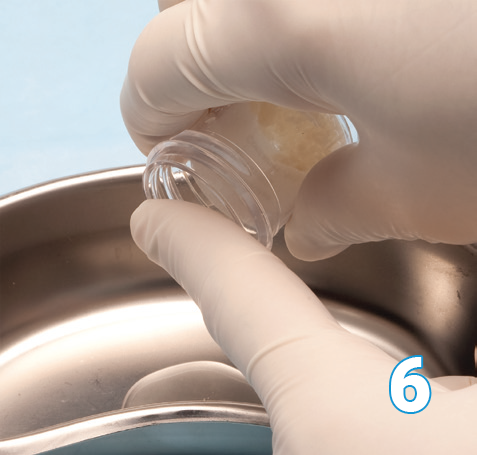
The cryopreservation cell saving liquid should be decanted and discarded.
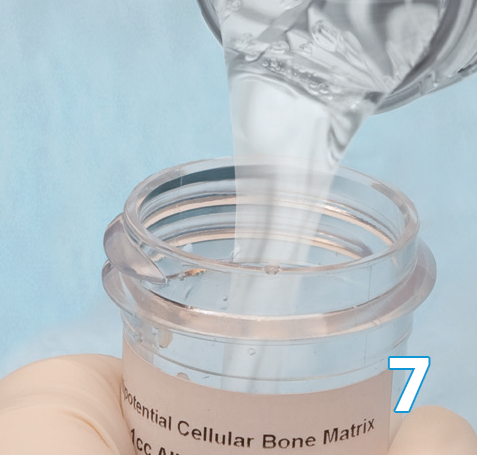
Add warm (95-102°F / 35-39°C) sterile saline to fully immerse Osteocel Plus. Osteocel® Plus can be maintained in saline for up to 4 hours.
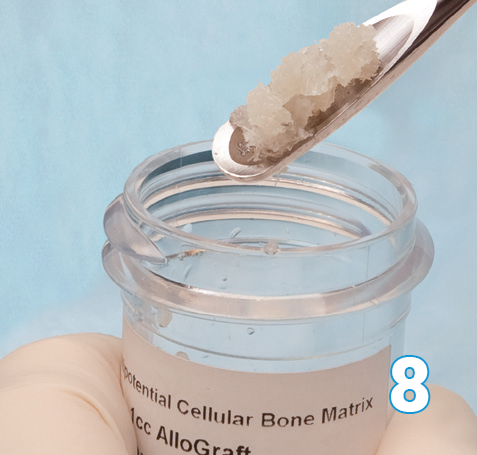
Pour off saline prior to use.
Note: Do not allow the cells to dry out.
Osteocel Plus is now ready for use.
1 Mesenchymal Stem Cells Avoid Allogeneic Rejection. – Ryan, Barry, Murphy, Mahon. – Journal of Inflammation, July 2005
2 Histologic Evaluation of a Stem Cell Based Sinus Augmentation Procedure: A Case Series. – McAllister, Haghighat, Gonshor. – Journal of Perio., April 2009
3 Data on file with NuVasive, Inc.
4 Data on file with Osiris Therapeutics
